support
Applications table
We cover a large variety of applications, discover them all below.

Autoclaves
Autoclaves are essential for the Life Sciences Industry. The validation of sterilization cycles is critical to ensure sterility of product, components and instruments, among others.
Wireless dataloggers are the best autoclave validation technology available today. Lives International’s XpertLog® wireless data loggers optimize the calibration and setup processes. They can also reduce by half the amount of labor needed to execute a validation cycle. These loggers feature 4-wire PT-100 sensors accurate to 0.1°C that significantly reduce the measurement uncertainty and improve the calculation of accumulated lethality, or F-value,over that of thermocouple-based systems. The XpertLog® software calculates the accumulated lethality delivered in a cycle and compares results to acceptance criteria easily configured following applicable standards such as EN 285 and ISO 17665 to produce a pass or fail result. This reduces human error in the interpretation of results and ensures data integrity.
The presence of saturated steam is crucial to ensure the efficacy of a moist heat sterilization cycle. When a XpertLog® pressure logger is used, our software can calculate and report whether the steam used in a sterilization cycle achieved saturation.
Lives International offers a wide range of dataloggers and accessories to suit your specific autoclave validation needs.
Contact us today to assist you with the best solution for your thermal validation needs.
Temperature data logger with flexible sensor

Standard data logger GMP F
Temperature and pressure data logger with rigid sensor

Standard data logger GMP TP
Real time wireless temperature data logger with 5 flexible sensors

Wireless data logger GMP RF 5F
Real time wireless temperature data logger with rigid sensor

Wireless data logger GMP RF
Temperature data logger low profile with flexible sensor

Standard data logger GMP F LP
Temperature and pressure data logger with flexible sensor

Standard data logger GMP TP F
High temperature real time wireless data logger with 5 flexible sensor

Wireless data logger GMP RF 5F HT
Real time temperature and pressure data logger with rigid sensor

Wireless data logger GMP RF TP
Stopper washer temperature data logger with rigid sensor

Standard data logger GMP SW
Real time wireless temperature data logger with flexible sensor

Wireless data logger GMP RF F
Real time wireless temperature data logger with flexible sensor

Wireless data logger GMP RF F 10RA
Real time wireless temperature and pressure with flexible sensor

Wireless data logger GMP RF F TP
Temperature data logger with rigid sensor

Standard data logger GMP
Real time wireless data logger low profile with flexible sensor

Wireless data logger GMP RF F LP
Real time wireless temperature data logger with flexible sensor

Wireless data logger GMP RF F 30H
Matryoshka real time wireless temperature data logger with flexible sensor

Wireless data logger GMP RF F MTR

Lyophilizers (Freeze-Dryers)
The extreme low temperatures and limited shelf clearance in lyophilizers make thermal mapping with conventional equipment rather difficult. That is why we developed the GMPF LP RF low-profile temperature loggers.
When fitter with Shelf Mapping Caps, these data loggers can accurately transmit shelf temperature in real-time. Their low profile also provides plenty of clearance to support stopper closure applications. Wireless data loggers can reduce by half the amount of labor needed to setup a mapping cycle. Check our home page for the video “Data loggers vs. Thermocouples” for more information.
Lives International has also developed specialized data loggers for continuous flow lyophilizers. Our Lyo Caterpillar follows the process flow of a 10 ml or 30 ml vial while wirelessly transmitting real-time temperature data to the XpertLog® software for analysis.
Our sales team can assist you with the best solution for your thermal validation needs. Contact us today.
Temperature data logger low profile with flexible sensor

Standard data loggers GMP F LP
Real time wireless temperature data logger with flexible sensor

Wireless data logger GMP RF F 30H
Real time wireless temperature data logger with flexible sensor

Wireless data logger GMP RF F 20R8V
Temperature data logger low profile with flexible sensor

Standard data logger GMP RF F LP
Matryoshka real time wireless temperature data logger with flexible sensor

Wireless data logger GMP RF F MTR

Steam in place (SIP)
The span of large steam in place systems can make the installation of thermal validation thermocouples very difficult, if not impractical. Wireless data loggers can provide a practical alternative for this application due to the simplicity of their installation.
The GMP Pipe adapter can fix a data logger sensor inside a pipe to make accurate in-line measurements. Specialty brackets can hold the data loggers close to the measurement point. WHere needed, a Range Extender or repeater increases the coverage area of the data acquisition Access Point to support the validation of large steam in place systems. Altogether, the XpertLog system can reduce setup time significantly while providing an accurate lethality calculation.
Our sales team can assist you with the best solution for your thermal validation needs. Contact us today.
Temperature data logger with flexible sensor

Standard data loggers GMP F
Temperature and pressure data logger with rigid sensor

Standard data logger GMP TP
Real time wireless temperature data logger with 5 flexible sensors

Wireless data logger GMP RF 5F
Real time wireless temperature and pressure data logger with rigid sensor

Wireless data logger GMP RF TP
Temperature data logger low profile with flexible sensor

Standard data logger GMP F LP
Temperature and pressure data logger with flexible sensor

Standard data logger GMP TP F
Real time wireless temperature and pressure data logger with flexible sensor

Wireless data logger GMP RF F TP
Matryoshka real time wireless temperature data logger with flexible sensor

Wireless data logger GMP RF F MTR

Stability chambers
ICH Q1A provides guidance for the stability testing of new drug substances and products.
Lives International’s GDP relative humidity loggers deliver standard data logger capabilities suitable for testing related to the climatic zones covered by ICH Q1A. The GMP relative humidity loggers provide high relative humidity accuracy and an expanded temperature range. The GMP RF relative humidity loggers add real-time wireless data capabilities useful when performing open door testing, recovery testing and power failure applications.
Temperature and humidity data logger with rigid sensor

Standard data logger GDP TH
Temperature and humidity data logger with rigid sensor

Standard data logger GMP TH
Real time wireless temperature data logger with flexible sensor

Wireless data logger GMP RF F
Real time wireless temperature and humidity data logger with flexible sensor

Wireless data logger GMP RF TH F
Temperature data logger with flexible sensor

Standard data logger GMP F
Temperature and humidity data logger with flexible sensor

Standard data logger GMP TH F
Real time wireless temperature data logger low profile with flexible sensor

Wireless data logger GMP RF F LP
Real time wireless temperature and pressure data logger with rigid sensor

Wireless data logger GMP RF TP
Temperature data logger low profile with flexible sensor

Standard data logger GMP F LP
Temperature and pressure data logger with rigid sensor

Standard data logger GMP TP
Real time wireless temperature data logger with rigid sensor

Wireless data logger GMP RF
Real time wireless temperature and pressure data logger with flexible sensor

Wireless data logger GMP RF TP F

Cold rooms
Cold room mapping is a simple application that can be handled with various technologies. The XpertVal thermocouple-based system is a cost-effective solution for applications having many test points in close proximity.
It is particularly useful when real-time data is needed at a fast rate. The GDP temperature loggers provide a standard data logger capabilities for large cold rooms. They reduce personnel exposure time to cold temperatures due to their ease of installation. The GMP temperature loggers provide an expanded temperature range. The GMP RF temperature loggers add real-time wireless data capabilities useful when performing open door testing, recovery testing and power failure simulations.
All our temperature data collection systems can be pre-calibrated and post-verified in-site using dry blocks and traceable reference probes. Also, the analysis tools included in the XpertLog® software make it easy to apply pre-configured acceptance criteria to a data set to oobtain a pass or fail result. This reduces human error in the interpretation of results and ensures data integrity. The statistic report automatically identifies the coldest and warmest locations from the mapping data. The software can also calculate kinetic temperature MKT, when applicable. All our thermal validation systems use the XpertLog® software which our customers rely upon their most critical applications.
Temperature and humidity data logger with rigid sensor

Standard data logger GDP TH
Temperature data logger with rigid sensor

Standard data logger GMP
Real time wireless temperature data logger low profile with flexible sensor

Wireless data logger GMP RF F LP
Temperature data logger with flexible sensor

Standard data logger GDP F
Temperature and humidity data logger with rigid sensor

Standard data logger GMP TH
Real time wireless temperature data logger with 5 flexible sensors

Wireless data logger GMP RF 5F

Sterilization tunnels
Sterilization tunnels (depyrogenation tunnels) are important in ensuring pharmaceutical equipment is sterile by moving equipment through a high-heat tunnel at a predetermined rate.
These tunnels must be validated periodically to ensure the heat inside is at the rate it should be.
The thermal mapping of continuous processing sterilization tunnels can be best achieveed using real time data loggers. These can easily be positioned in the process flow to record temperature throughout the different process stages.
One of the challenges in validating the temperature inside deypyrogenation tunels -besides the high temperature inside – is the fact that the measuring equipment must be inserted at one end of the tunnel and gets retrieved at the other end. Very often this fact generates delays and often damages the equipment.
To address this issue we designed a high temperature data logger that wirelessly transmits real time data from inside of the sterilization tunnel. This high temperature data logger can withstand up to +350°C and can be easily used by introducing it at one end of the tunnel and retrieve it at the other end.

Ovens
Ovens are widely used for drying, warming, sterilization, melting, baking various sub-products, parts, substances or final products and they must maintain the specified temperature.
The XpertVal is a thermocouple-based thermal validation system that is suitable to handle the high temperatures typically encountered in ovens. The technology has the capability to record temperature data for up to 120 test points in real-time.
For oven temperatures below 140°C, GMP temperature loggers provide a higher accuracy of 0.1°C and easier installation that reduces labor cost. The GMP RF temperature loggers add real-time wireless data capabilities.
Both systems are supported by the XpertLog software® which our customers rely upon for their most critical applications.

Freezers
Freezer temperatures may vary and typically range from -25°C to -10°C. Regulations require that these systems be periodically mapped to ensure that they continue to fulfill their intended purpose.
Mapping can be performed using various technologies. The XpertVal thermocouple-based system is a cost-effective soltuion. It is particularly useful when real-time data is needed at a fast rate. it also coves the widest temperature range of all systems. The GDP temperature loggers provide standard data logger capabilities, simplifying installation and reducing the associated labor. The GMP temperature loggers provide an expanded temperature range suitable for low-temperature applications. The GMP RF temperature loggers add real-time wireless data capabilities useful when performing open door testing, recovery testing and power failure simulations.
All our temperature data collection systems can be pre-calibrated and post-verified in-site using dry blocks and traceable reference probes. Also, the analysis tools included in the XpertLog® software make it easy to apply pre-configured acceptance criteria to a data set to obtain a pass or fail result. This reduces human error in the interpretation of results and ensures data integrity. The statistics report automatically indentifies the coldest and warmest locations from the mapping data. The software can also calculate kinetic temperature MKT, when applicable. All our thermal validation systems use the XpertLog® software which our customers rely upon for their most critical applications.
Temperature data logger with flexible sensor

Standard data logger GMP F
Real time wireless temperature data logger low profile with flexible sensor

Standard data logger GMP RF F LP
Temperature data logger low profile with flexible sensor

Standard data logger GMP F LP
Real time wireless temperature data logger with 5 flexible sensors

Wireless data logger GMP RF 5F

Refrigerators
The reliability of refrigerators is crucial for pharmaceutical and biotech companies, as temperature-sensitive products are stored in cold environments. Refrigerators used for these purposes must be periodically validated to ensure they are keeping products at the proper temperature no matter where in the refrigerator they are placed.
Temperature data logger with flexible sensor

Standard data logger GDP F
Real time wireless data logger with flexible sensor

Wireless data logger GMP RF F
Temperature data logger with flexible sensor

Standard data logger GMP F
Real time wireless data logger low profile with flexible sensor

Wireless data logger GMP RF F LP

Incubators
Incubator mapping can be easily performed with a combination of temperature and CO2 data loggers. The GMP RF CO2 RF data logger records and transmits temperature and CO2 data.
Additional GMP RF loggers can complement temperature distribution data, all in real-time.
Our GDP and GMP temperature loggers could also provide the temperature distribution data with high accuracy. All our thermal validation systems use the XpertLog® software wich our customers rely upon for their most critical applications.
Temperature and humidity data logger with rigid sensor

Standard data logger GDP TH
Temperature data logger with rigid sensor

Standard data logger GMP
Real time wireless temperature data logger low profile with flexible sensor

Wireless data logger GMP RF F LP
Real time wireless temperature and CO2 data logger with flexible sensor

Wireless data logger GMP RF TCO2 F
Temperature data logger with flexible sensor

Standard data logger GDP F
Temperature and humidity data logger with rigid sensor

Standard data logger GMP TH
Real time wireless temperature data logger with rigid sensor

Wireless data logger GMP RF

Cold chain shipments
Tracking the temperature of a product during cold chain shipping is a problem for many in the pharmaceutical industry. If insulated containers fail to properly keep a product at its designated temperatures, the product may be ruined by the time it arrives. When this happens, it is costly and a waste of time.
Ensure that your products are kept at the perfect temperature with the XperTrack sytem from Lives International. The XperTrack system allows you to see the temperature and location of a product in real-time during shipment, allowing for quick correction if the product goes outside of the desired temperature range. This system is wireless and easy to use, as all you need to do is place a temperature logger inside the container with your product before shipping.
Temperature and humidity data logger with rigid sensor

Standard data logger GDP TH
Temperature data logger with rigid sensor

Standard data logger GMP
Real time wireless temperature data logger low profile with flexible sensor

Wireless data logger GMP RF F LP
Real time wireless temperature data logger with rigid sensor

Wireless data logger GMP RF
Temperature data logger with flexible sensor

Standard data logger GDP F
Temperature and humidity data logger with rigid sensor

Standard data logger GMP TH
Real time wireless temperature data logger with 5 flexible sensors

Wireless data logger GMP RF 5F

Cold chain shipments
Wireless data loggers are ideal for warehouse mapping since they are very easy to install. Our GDP humidity and temperature loggers provide standard data logger capabilities.
The GMP humidity and temperature loggers provide higher relative humidity accuracy where tighter tolerances are called for. The real time wireless capability of GMP RF loggers alert about any humidity or temperature excursions early in the mapping process allowing to proactively implement corrective actions.
Check our blog for recommendations on how to determine the number of sensors needed for warehouse mapping.
Temperature and humidity data logger with rigid sensor

Standard data logger GDP TH
Temperature data logger with rigid sensor

Standard data logger GMP
Real time wireless temperature data logger low profile with flexible sensor

Wireless data logger GMP RF F LP
Temperature data logger with flexible sensor

Standard data logger GDP F
Temperature and humidity data logger with rigid sensor

Standard data logger GMP TH
Real time wireless temperature data logger with rigid sensor

Wireless data logger GMP RF

Shipping studies
Shipping studies are needed to evaluate whether the shipping process meets specified product storage conditions. Our GDP temperature and humidity loggers are ideal for this task.
They can fit in small spaces. They provide ample memory capacity to record temperature and humidity data for long shipment durations. These loggers are easy to set up and download in batches.
Our XpertLog software effortlessly compiles logger data into a single meaningful report. Also, the analysis tools included in the software make it easy to apply pre-configured acceptance criteria to a data set to obtain a pass or fail result. This reduces human error in the interpretation of results and ensures data integrity. The statistics report automatically indentifies the coldest and warmest locations from the mapping data. The software can also calculate mean kinetic temperature MKT, when applicable.
Contact us today to assist you with the best solution for your requirements.

Package integrity
Packaging your products requires that they stay at the right temperature and humidity during shipment. When the product is in transit, you don’t know if there will be a tear or leak in the seal, running the product inside the package. With Lives International, you can monitor package integrity during all points of shipment.
When preparing your product for shipment, simply place one of the wireless data loggers into the package along with your product. The logger will send temperature data to a cloud, which you can access from any internet-connected device. The data will constantly update during transit, and you will be sent an alert if the temperature goes outside a defined range.
Learn more about package integrity control with XperTrack today.
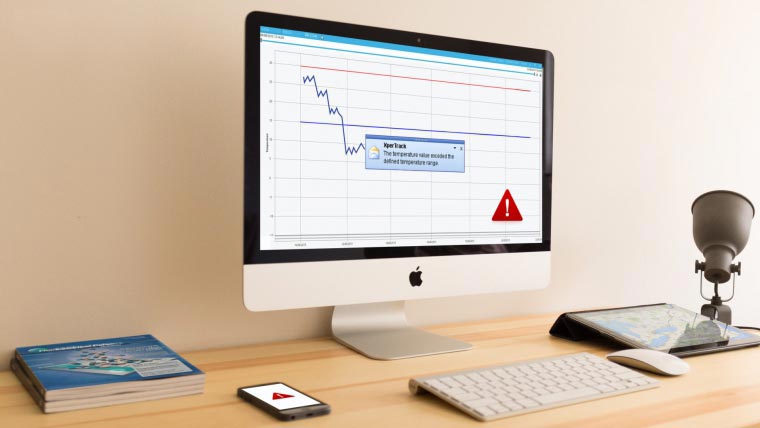
Remote monitoring
When you’re shipping your temperature-sensitive product, you are aware of all the things that can go wrong. The product can be too warm or cold, be dropped and broken, or be lost altogether. The solution for all of these problems is remote monitoring for cold chain shipments, such as the XperTrack system from Lives International.
The XperTrack system allows you to monitor your products’ temperature, shocks and location during cold chain shipment. With this remote monitoring system, you’ll be able to see information about your shipments in real time from any internet-connected device. You’ll also receive alerts in case the temperature goes outside a defined limit, or in case the product registers a larg shock such as a drop.
Learn more about how the XperTrack system is perfect for your cold chain remote monitoring by contacting Lives International today.

Microbial detection systems
We created the GMP RF Bio product especially for use inside the BacT/Alert 3D microbial detection system.
BacT/Alert 3D is used for sepsis and quality control testing, to monitor for the presence or absence of microorganisms.
The GMP RF Bio data loggers have the same size and shape as the vials used inside and allow you to validate the BacT/Alert equipment.

Rubber stopper washing machine
Rubber stoppers washing machines are widely used in pharma industry and they require validation of the washing/sterylization cycle. Most of the stopper washers available today compine multiple features, like washing, sterilizing, hot steam drying and siliconizing rubber stoppers, aluminium caps, plungers, all kinds of disk seals and closures.
The GMP SW temperature data loggers are specially designed to be used in rubber stopper washing machine. GMP SW records a temperature range between -40°C to +140°C and can also be used in autoclaves or other types of equipment that require high temperature and humidity resistant data loggers. The temperature sensor is incorporated in the data logger body, allowing temperature values to be recorded with high accuracy (±0.1°C) while resisting shock and friction inside the rubber stopper washing machines.

Logistics
Wireless data loggers are ideal for warehouse mapping since they are very easy to install. Our GDP humidity and temperature loggers provide standard data logger capabilities.
The GMP humidity and temperature loggers provide higher relative humidity accuracy where tighter tolerances are called for. The real time wireless capability of GMP RF loggers alert about any humidity or temperature excursions early in the mapping process allowing to proactively implement corrective actions.
Check our blog for recommendations on how to determine the number of sensors needed for warehouse mapping.
Temperature and humidity data logger with rigid sensor

Standard data logger GDP TH
Temperature data logger with rigid sensor

Standard data logger GMP
Real time wireless temperature data logger low profile with flexible sensor

Wireless data logger GMP RF F LP
Real time wireless temperature data logger low profile with 5 flexible sensors

Wireless data logger GMP RF 5F
Temperature data logger with flexible sensor

Standard data logger GDP F
Temperature and humidity data logger with rigid sensor

Standard data logger GMP TH
Real time wireless temperature data logger with rigid sensor

Wireless data logger GMP RF

Ultra low freezers
Ultra low freezers or ULT freezers are typically used to store contents between -40°C to -86°C. ULT freezers are used for long term storage of proteins, cell extracts or reagents, and thus they require periodic validation.
Lives International produces a wide range of both standard and wireless real time data loggers, with various shapes and sizes, that work down to -90°C.
Temperature data logger with rigid sensor

Standard data logger GMP
Real time wireless temperature data logger low profile with flexible sensor

Wireless data logger GMP RF F LP
Temperature data logger with flexible sensor

Standard data logger GMP F
Real time wireless temperature data logger low profile with 5 flexible sensors





